FDA OKs new therapy for some hemophilia patients
U.S. regulators have approved the first new treatment in nearly two decades to prevent internal bleeding in certain patients with hemophilia.
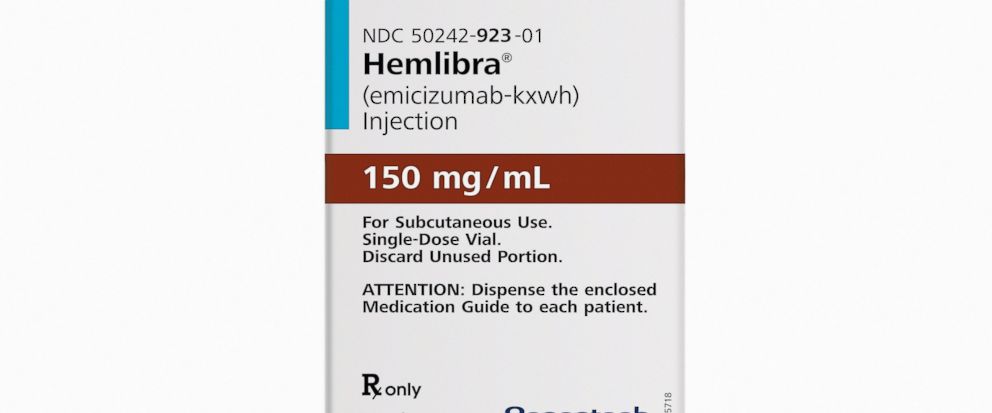
The Food and Drug Administration on Thursday approved Hemlibra, a weekly self-injection for hemophilia A patients who have developed resistance to standard medicines for preventing bleeding episodes.
The list price will be about $482,000 for the first year and slightly less after that, said California-based Genentech, which developed the drug.
Genentech, the biotech subsidiary of Swiss drugmaker Roche, says that's half the cost of the only other preventive option for patients with this problem. That treatment requires a two-hour IV drip several times a week.
About 20,000 Americans — mostly males — have hemophilia, an inherited, potentially life-threatening disorder. Their blood doesn't clot properly because of a faulty gene. In severe cases, repeated bleeding in the joints leads to problems walking and intense pain. About 80 percent of hemophilia patients have hemophilia A and about one-third of those develop resistance.
The FDA gave expedited approval to Hemlibra, also known as emicizumab, for adults and children who have that resistance. Genentech continues to test Hemlibra, with the aim of getting it approved for all hemophilia A patients.
In a company-funded study, participants who got Hemlibra had a 79 percent reduction in the number of bleeding episodes compared to how they had previously fared on the older treatment. The FDA noted the patients getting Hemlibra reported less pain, joint swelling and difficulty walking.
Hemlibra carries the FDA's strongest level of warning because it can cause severe blood clots when patients are given a certain other bleeding treatment at the same time. Common side effects include headache, joint pain and injection site reactions.
Greg Hogan's son, Patrick, was diagnosed with hemophilia after birth. At 9 months, Patrick developed such severe resistance to his medicine that he had to get massive quantities through an IV every morning. Still, the family had to severely limit his activity including playing with his brothers. They also made him wear a helmet and padded the interior of their home in Kathleen, Georgia, with protective foam.
Patrick started receiving Hemlibra nine months ago as part of the testing. His father said he improved immediately, feeling stronger and behaving "more like a rambunctious child." Patrick, now 4, hasn't had a bleeding episode since starting on the treatment, not even after twisting his ankle.
"That would have been extremely serious" before, said Hogan, who has taken down the foam padding and now often lets Patrick leave his helmet off. "I would let him play any sport except football."
———
Follow Linda A. Johnson at @LindaJ—onPharma
- Star